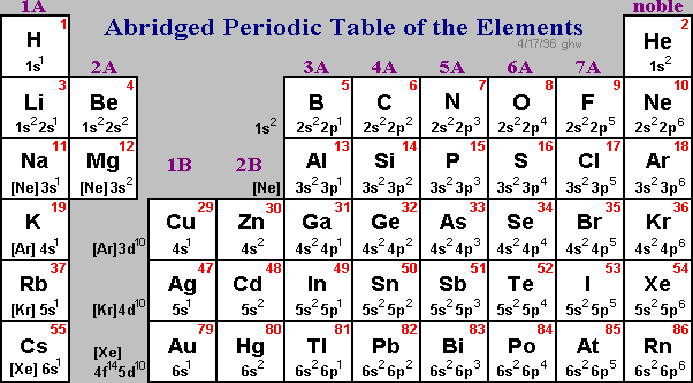
Introduction to the Periodic Table
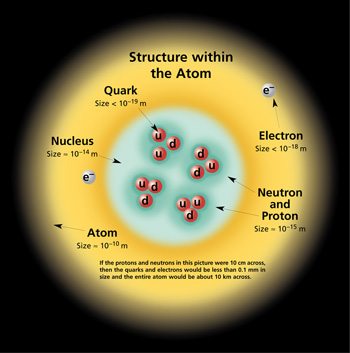
Scientists use the Periodic Table in order to find out important information about various elements. Created by Dmitri Mendeleev (1834-1907), the periodic table orders all known elements in accordance to their similarities. When Mendeleev began grouping elements, he noticed the Law of Chemical Periodicity. This law states, "the properties of the elements are periodic functions of atomic number." The periodic table is a chart that categorizes elements by "groups" and "periods." All elements are ordered by their atomic number. The atomic number is the number of protons per atom. In a neutral atom, the number of electrons equals the number of protons. The periodic table represents neutral atoms. The atomic number is typically located above the element symbol. Beneath the element symbol is the atomic mass. Atomic mass is measured in Atomic Mass Units where 1 amu = (1/12) mass of carbon measured in grams. The atomic mass number is equal to the number of protons plus neutrons, which provides the average weight of all isotopes of any given element. This number is typically found beneath the element symbol. Atoms with the same atomic number, but different mass numbers are called isotopes. Below is a diagram of a typical cells on the periodic table.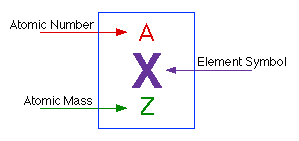
There are two main classifications in the periodic table, "groups" and "periods." Groups are the vertical columns that include elements with similar chemical and physical properties. Periods are the horizontal rows. Going from left to right on the periodic table, you will find metals, then metalloids, and finally nonmetals. The 4th, 5th, and 6th periods are called the transition metals. These elements are all metals and can be found pure in nature. They are known for their beauty and durability. The transition metals include two periods known as the lanthanides and the actinides, which are located at the very bottom of the periodic table. The chart below gives a brief description of each group in the periodic table.
| Group 1A |
|
| Group 2A |
|
| Group 3A |
|
| Group 4A |
|
| Group 5A |
|
| Group 6A |
|
| Group 7A |
|
| Group 8A |
|
Charges in the Atom
The charges in the atom are crucial in understanding how the atom works. An electron has a negative charge, a proton has a positive charge and a neutron has no charge. Electrons and protons have the same magnitude of charge. Like charges repel, so protons repel one another as do electrons. Opposite charges attract which causes the electrons to be attracted to the protons. As the electrons and protons grow farther apart, the forces they exert on each other decrease.Atomic Models and the Quantum Numbers
There are different models of the structure of the atom. One of the first models was created by Niels Bohr, a Danish physicist. He proposed a model in which electrons circle the nucleus in "orbits" around the nucleus, much in the same way as planets orbit the sun. Each orbit represents an energy level which can be determined using equations generated by Planck and others discussed in more detail below. The Bohr model was later proven to be incorrect, but provides a useful model for building an explanation.The "accepted" model is the quantum model. In the quantum model, we state that the electron cannot be found precisely, but we can predict the probability, or likelihood, of an electron being at some location in the atom. You should be familiar with quantum numbers, a series of three numbers used to describe the location of some object (like an electron) in three-dimensional space:- n: the principal quantum number, an integer value (1, 2, 3...) that is used to describe the quantum level, or shell, in which an electron resides. The principal quantum number is the primary number used to determine the amount of energy in an atom. Using one of the first important equations in atomic structure (developed by Niels Bohr), we can calculate the amount of energy in an atom with an electron at some value of n:
En = - Rhc
n2
R = Rydberg constant, a value of 1.097 X 107 m-1
c = speed of light, 3.00 X 108 m/s
h = Planck's constant, 6.63 X 10 -34 J-s
n = principal quantum number, no unitFor example, how much energy does one electron with a principal quantum number of n= 2 have?
En = - Rhc
n2or En = - (1.097x107 m-1 ∗ (6.63x10-34 J•s)∗(3.0x108 m•s-1)
22= 5.5x10-19 J
ΔE = hv = RH 1
ni2- 1
nf2
ΔE = change in energy (Joules)
h = Planck's constant with a value of 6.63 x 10-34 (J-s)
ν is frequency (s-1)
RH is the Rydberg constant with a value of 2.18 x 10-18J.
ni is the initial quantum number
nf is the final quantum number
Using the equation below, we can calculate the wavelength and the frequency of the energy. The wavelength and the frequency give us information about how we might "see" the energy:
vλ = c
ν = the frequency of radiation (s-1)
λ = the wavelength (m)
c = the speed of light with a value of 3.00 x 108 m/s in a vacuum
Speed of light = 3.00E+08 Rydberg constant = 2.18E-18 Planck's constant = 6.63E-34 Excited state, n = 3 4 5 Ground state, n = 2 2 2 Excited state energy (J) 2.42222E-19 1.363E-19 8.72E-20 Ground state energy (J) 5.45E-19 5.45E-19 5.45E-19 ΔE = -3.02778E-19 -4.09E-19 -4.58E-19 ν = 4.56678E+14 6.165E+14 6.905E+14 λ(nm) = 656.92 486.61 434.47
- l ("el", not the number 1): the azimuthal quantum number, a number that specifies a sublevel, or subshell, in an orbital. The value of the azimuthal quantum number is always one less than the principal quantum number n. For example, if n=1, then "el"=0. If n=3, then l can have three values: 0,1, and 2. The values of l are typically not identified as "0, 1, 2, and 3" but are more commonly called by their historic names, "s, p, d, and f", respectively. Since the quantum numbers were discovered through the study of light and lines on an electromagnetic spectra, chemists identified the lines by their quality:sharp, principal, diffuse and fundamental. The table below shows the relationship:
Value of l Subshell designation 0 s 1 p 2 d 3 f - m: the magnetic quantum number. Each subshell is composed of one or more orbitals. In the study of light, it was discovered that additional lines appeared in the spectra produced when light was emitted in a magnetic field. The magnetic quantum number has values between -l and +l. When l =1, for example, m can have three values: -1, 0, and +1. Because you know from the chart above that the subshell designation for l =1 is "p", you now know that the p orbital has three components. In your study of chemistry, you will be presented with px, py, and pz. Notice how the subscripts are related to a three-dimensional coordinate system, x, y, and z. The chart below shows a summary of the quantum numbers:
Principal Quantum Number (n) Azimuthal Quantum Number (l) Subshell Designation Magnetic Quantum Number (m) Number of orbitals in subshell 1 0 1s 0 1 2 0
12s
2p0
-1 0 +11
33 0
1
23s
3p
3d0
-1 0 +1
-2 -1 0 +1 +21
3
54 0
1
2
34s
4p
4d
4f0
-1 0 +1
-2 -1 0 +1 +2
-3 -2 -1 0 +1 +2 +31
3
5
7
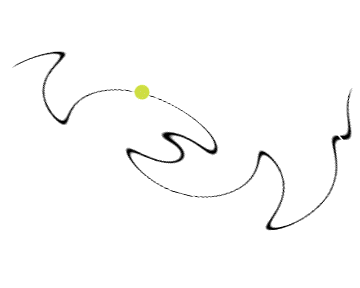
Chemists care about where electrons are in an atom or a molecule. In the early models, we believed that electrons move like billiard balls, and followed the rules of classical physics. The graphic below attempts to show that earlier models thought that we could identify the exact path, position, velocity, etc. of an electron or electrons in an atom:

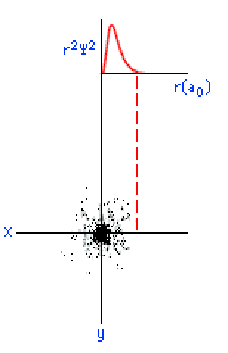
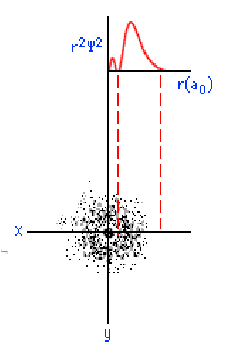
The three graphics below show some other orbitals. The first graph (top left) is of a "2s" orbital. Each "s" orbital can hold two electrons in its cloud. Notice how there is a relatively high probability of an electron being near the nucleus, then some space where the probability is close to zero, then the probability increases substantially at some distance from the nucleus. The graphic at the top right shows a "2p" atomic orbital. Orbitals that are "p" orbitals can hold up to six (6) electrons in their cloud. Notice its "dumbbell" or "figure of eight" shape. At the bottom left is a "3s" orbital. Again, notice its spherical shape. Finally, at the bottom right, is a "3p" orbital.
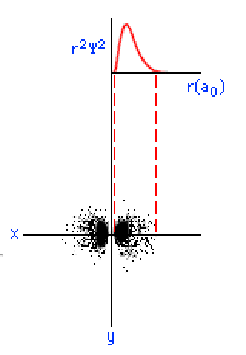
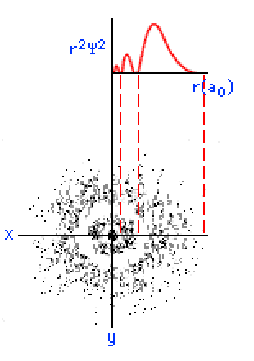
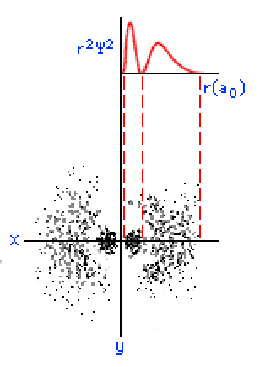
Determining Electron Configuration
One of the skills you will need to learn to succeed in freshman chemistry is being able to determine the electron configuration of an atom. An electron configuration is basically an account of how many electrons there are, and in what orbitals they reside under "normal" conditions. For example, the element hydrogen (H) has one electron. We know this because its atomic number is one (1), and the atomic number tells you the number of electrons. Where does this electron go? The one electron of hydrogen goes into the lowest energy state it possibly can, which means it will start at "level" one and goes into "s" orbitals first. We say that hydrogen has a "[1s1]" electron configuration. Looking at the next element on the Periodic Table --helium, or He -- we see it has an atomic number of two, so two electrons. Since " s" orbitals can hold up to two electrons, helium has an electron configuration of "[1s2]".What about larger atoms? Let's look at carbon, with an atomic number of 6. Where do its 6 electrons go?- First two: 1s2
- Next two: 2s2
- Last two: 2p2
The table below shows the subshells, the number of orbitals, and the maximum number of electrons allowed:
| Subshell | Number of Orbitals | Maximum Number of Electrons |
|---|---|---|
| s | 1 | 2 |
| p | 3 | 6 |
| d | 5 | 10 |
| f | 7 | 14 |
Try It Out
- What is the frequency of infrared radiation that has a wavelength of 1.25 x 103 nm?Using the equation:
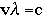
where:
v = frequency (in units of "per second", or s-1
c = speed of light at 3.0 x 108 m/s
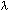 = wavelength in units of nanometers (nm)
= wavelength in units of nanometers (nm)
Solving for frequency, we have the equation:
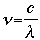
To ensure that our units match, we must convert the wavelength from nanometers to meters:
1.25 x 103 nm * (1 x 10-9 meters / 1 nm) = 1.25 x 10-6 m
Now, it's just a simple "plug and chug":
v = 3.00 x 108 m s-1 / 1.25 x 10-6 m
= 2.40 x 1014 s-1 or 2.40 x 1014 s-1 hertz (Hz)
INTERNATIONAL CONCEPT OF WORK FROM HOME
ReplyDeleteWork from home theory is fast gaining popularity because of the freedom and flexibility that comes with it. Since one is not bound by fixed working hours, they can schedule their work at the time when they feel most productive and convenient to them. Women & Men benefit a lot from this concept of work since they can balance their home and work perfectly. People mostly find that in this situation, their productivity is higher and stress levels lower. Those who like isolation and a tranquil work environment also tend to prefer this way of working. Today, with the kind of communication networks available, millions of people worldwide are considering this option.
Women & Men who want to be independent but cannot afford to leave their responsibilities at home aside will benefit a lot from this concept of work. It makes it easier to maintain a healthy balance between home and work. The family doesn't get neglected and you can get your work done too. You can thus effectively juggle home responsibilities with your career. Working from home is definitely a viable option but it also needs a lot of hard work and discipline. You have to make a time schedule for yourself and stick to it. There will be a time frame of course for any job you take up and you have to fulfill that project within that time frame.
There are many things that can be done working from home. A few of them is listed below that will give you a general idea about the benefits of this concept.
Baby-sitting
This is the most common and highly preferred job that Women & Men like doing. Since in today's competitive world both the parents have to work they need a secure place to leave behind their children who will take care of them and parents can also relax without being worried all the time. In this job you don't require any degree or qualifications. You only have to know how to take care of children. Parents are happy to pay handsome salary and you can also earn a lot without putting too much of an effort.
Nursery
For those who have a garden or an open space at your disposal and are also interested in gardening can go for this method of earning money. If given proper time and efforts nursery business can flourish very well and you will earn handsomely. But just as all jobs establishing it will be a bit difficult but the end results are outstanding.
Freelance
Freelance can be in different wings. Either you can be a freelance reporter or a freelance photographer. You can also do designing or be in the advertising field doing project on your own. Being independent and working independently will depend on your field of work and the availability of its worth in the market. If you like doing jewellery designing you can do that at home totally independently. You can also work on freelancing as a marketing executive working from home. Wanna know more, email us on workfromhome.otr214428@gmail.com and we will send you information on how you can actually work as a marketing freelancer.
Internet related work
This is a very vast field and here sky is the limit. All you need is a computer and Internet facility. Whatever field you are into work at home is perfect match in the software field. You can match your time according to your convenience and complete whatever projects you get. To learn more about how to work from home, contact us today on workfromhome.otr214428@gmail.com and our team will get you started on some excellent work from home projects.
Diet food
Since now a days Women & Men are more conscious of the food that they eat hence they prefer to have homemade low cal food and if you can start supplying low cal food to various offices then it will be a very good source of income and not too much of efforts. You can hire a few ladies who will help you out and this can be a good business.
Thus think over this concept and go ahead.